| 1822556920 | Explain concept 9.1: Catabolic pathways yield energy by oxidizing organic fuels | Catabolic pathways are a set of metabolic pathways that breaks down molecules into smaller units to release energy | | 0 |
| 1822556921 | Compare and contrast aerobic and anaerobic respiration | Both processes include glycolysis, the citric acid cycle, and oxidated phosphorylation. In aerobic respiration the final electron acceptor is molecular oxygen O2; in anaerobic respiration the final electron Accceptor is a different substance. | | 1 |
| 1822556922 | Describe the difference between the two processes in cellular respiration that produce ATP: oxidative phosphorylation and substrate-level phosphorylation | Most of the ATP produced in cellular respiration come from oxidative phosphorylation, in which the energy released from redox reactions in an electron transport chain is used to produce ATP.
In substrate level phosphorylation, an enzyme dirctly transfers a phosphate group to ADP fro an intermediate substrate. All ATP production in glycolysis occurs by substrate-level phosphorylation, this form of ATP production also occurs at one step in the citric acid cycle. | | 2 |
| 1822557023 | Explain concept 9.2: Glycolysis harvests chemical energy by oxidizing glucose to pyruvate | ... |  | 3 |
| 1822556923 | During the redox reaction in glycolysis, which molecule acts as the oxidizing agent? Which molecule acts as the reducing agent? | NAD+ acts as the oxidizing agent in step 6, accepting electrons from glyceraldehyde 3-phosphate, which thus acts as the reducing agent | | 4 |
| 1822556924 | During the redox reaction in glycolysis, Which molecule acts as the reducing agent? | glyceraldehyde 3-phosphate, which acts as the reducing agent | | 5 |
| 1822557024 | What is the source of energy for the formation of ATP and NADH in glycolysis? | The oxidation of the three carbon sugar glyceraldehyde 3-phosphate, yields energy. In this oxidation, electrons and H+ are transferred to NAD+, forming NADH, and a phosphate group is attached to the oxidized substrate. ATP is then formed b y substrate level phosphorylation when this phosphate group is transferred to ADP |  | 6 |
| 1822556925 | Explain concept 9.3: After pyruvate is oxidized, the citric acid cycle completes the energy yielding oxidation of organic molecules | ... | | 7 |
| 1822557025 | Name the molecules that conserve most of the energy from the citric acid cycle's redox reactions. How is this energy converted to a form that can be used to make ATP? | NADH and FADH2; they will donate electrons to the electron transport chain |  | 8 |
| 1822556926 | What processes in your cells produce the CO2 that you exhale | CO2 is released from the pyruvate that is the end product of glycoloysis, and CO2 is also released during the citric acid cycle | | 9 |
| 1822556927 | What molecular products indicate the complete oxidation of glucose during cellular respiration? | ... | | 10 |
| 1822556928 | Explain concept 9.4: During oxidative phosphorylation, chemiosmosis couples electron transport to ATP synthesis | ... | | 11 |
| 1822557026 | What effect would an absence of O2 have on the process shown in figure 9.15? | ... |  | 12 |
| 1822557027 | Briefly explain the mechanism by which ATP synthase produces ATP. List three locations in which ATP synthases are found. | ... |  | 13 |
| 1822557028 | Explain concept 9.5: Fermentation and anaerobic respiration enable cells to produce ATP without the use of oxygen | ... |  | 14 |
| 1822556929 | Consider the NADH formed during glycolysis. What is the final acceptor for its electrons during fermentation? | ... | | 15 |
| 1822556930 | Consider the NADH formed during glycolysis. What is the final acceptor for its electrons during aerobic respiration? | ... | | 16 |
| 1822556931 | Which process yields more ATP, fermentation, or anaerobic respiration? | ... | | 17 |
| 1822556932 | Explain concept 9.6: Glycolysis and the citric acid cycle connect to many other metabolic pathways | ... | | 18 |
| 1822556933 | Compare the structure of a fat with that of a carbohydrate, what features of their structure make fat a much better fuel? | ... | | 19 |
| 1822556934 | Under what circumstances might your body synthesize fat molecules | ... | | 20 |
| 1822556935 | Describe how catabolic pathways of glycolysis and the citric acid cycle intersect with anabolic pathways in the metabolism of a cell. | ... | | 21 |
| 1822556936 | As it pertains to cellular respiration, state which organisms undergo cellular respiration. | ... | | 22 |
| 1822556937 | As it pertains to cellular respiration, distinguish between the site of cellular respiration in prokaryotic cells and in eukaryotic cells. | ATP synthases are found in the inner mitochondrial membranes of eukaryotic cells and in the plasma membranes of prokaryotes. | | 23 |
| 1822556938 | As it pertains to cellular respiration, distinguish between the terms aerobic and anaerobic. | ... | | 24 |
| 1822556939 | As it pertains to cellular respiration, write the general chemical equation for aerobic cellular respiration. | ... | | 25 |
| 1822556940 | As it pertains to cellular respiration, write the general chemical equation for lactic acid fermentation and state which organisms can undergo this process. | ... | | 26 |
| 1822556941 | As it pertains to cellular respiration, write the general chemical equation for alcohol fermentation and state which organisms can undergo this process. | ... | | 27 |
| 1822556942 | Relating to cellular energy: explain the chemical energy relationship between glucose and ATP. | ... | | 28 |
| 1822556943 | Relating to cellular energy: explain the chemical energy role of ATP in driving cellular reactions. | ... | | 29 |
| 1822556944 | Relating to cellular energy: describe the structure of ATP, ADP, and AMP. | ... | | 30 |
| 1822556945 | Relating to cellular energy: explain why ATP is considered the "energy currency" of the cell and glucose is not. | ... | | 31 |
| 1822556946 | Relating to cellular energy: state how many ATPs are produced from 1 glucose molecule during aerobic cellular respiration in prokaryotic cells | ... | | 32 |
| 1822556947 | Relating to cellular energy: state how many ATPs are produced from 1 glucose molecule during aerobic cellular respiration in eukaryotic cells | ... | | 33 |
| 1822556948 | Relating to cellular energy: state how many ATPs are produced from 1 glucose molecule during fermentation | ... | | 34 |
| 1822556949 | Describe the making of ATP through substrate-level phosphorylation. | ... | | 35 |
| 1822556950 | Describe how organic molecules other than glucose (specifically proteins, fats, and nucleic acids) can be a source of energy by being broken down and used during glycolysis, the transition reaction, or the Krebs cycle. | ... | | 36 |
| 1822557029 | Relating to oxidative phosphorylation (electron transport chain + chemiosmosis): define oxidation and reduction and describe the redox reactions of an electron transport chain. | ... |  | 37 |
| 1822556951 | Relating to oxidative phosphorylation (electron transport chain + chemiosmosis): relate the redox reactions of an electron transport chain to the active transport of
hydrogen ions (H+) across a membrane. | ... | | 38 |
| 1822556952 | Relating to oxidative phosphorylation (electron transport chain + chemiosmosis): relate the active transport of H+ ions across a membrane to the formation of an electrochemical gradient. | ... | | 39 |
| 1822557030 | Relating to oxidative phosphorylation (electron transport chain + chemiosmosis): relate the electrochemical gradient to the facilitated diffusion of H+ ions across a
membrane. | ... |  | 40 |
| 1822557031 | Relating to oxidative phosphorylation (electron transport chain + chemiosmosis): relate facilitated diffusion of H+ ions through the ATP synthase protein channel to
the making of ATP. | ... |  | 41 |
| 1822556953 | Relating to aerobic cellular respiration: describe the molecules ATP and NADH and distinguish between the different energy-storing roles of each. | ... | | 42 |
| 1822557032 | Relating to aerobic cellular respiration: describe the process of glycolysis, including the major molecules involved and the energy-storing molecules produced. | ... | 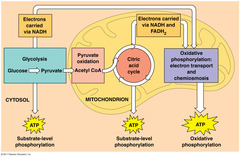 | 43 |
| 1822556954 | Relating to aerobic cellular respiration: describe the process of the transition reaction, including the major molecules involved and the energy-storing molecules produced. | ... | | 44 |
| 1822557033 | Relating to aerobic cellular respiration: describe the process of the Krebs cycle, including the major molecules involved and the energy-storing molecules produced, and explain why the Krebs cycle is considered a cycle. | ... |  | 45 |
| 1822557034 | Relating to aerobic cellular respiration: describe the roles of NADH, FADH2, and O2 in the electron transport chain part of oxidative phosphorylation | ... |  | 46 |
| 1822557035 | Relating to aerobic cellular respiration: show the relationship between the electron transport chain, active transport, and facilitated diffusion in the oxidative phosphorylation process of making ATP. | ... |  | 47 |
| 1822556955 | Relating to aerobic cellular respiration: state the specific locations of glycolysis, the transition reaction, the Krebs cycle, and the oxidative-phosphorylation process in eukaryotic cells. | ... | | 48 |
| 1822556956 | Relating to aerobic cellular respiration: state the specific locations of glycolysis, the transition reaction, the Krebs cycle, and the oxidative-phosphorylation process in prokaryotic cells. | ... | | 49 |
| 1822557036 | Relating to aerobic cellular respiration: state the number of ATPs produced during glycolysis, the transition reaction, the Krebs cycle, and the oxidative-phosphorylation process. | ... |  | 50 |
| 1822556957 | Relating to aerobic cellular respiration: explain why aerobic cellular respiration results in 36 ATPs per glucose in eukaryotic cells and 38 ATPs per glucose in prokaryotic cells. | ... | | 51 |
| 1822556958 | Relating to aerobic cellular respiration: relate glycolysis to lactic acid fermentation and alcohol fermentation. | ... | | 52 |
| 1822557037 | Fermentation | Partial degradation of sugars or other organic fuel that occurs without the use of oxygen |  | 53 |
| 1822557038 | Aerobic Respiration | Catabolic pathway in which oxygen is consumed as a reactant along with the organic fuel |  | 54 |
| 1822557039 | Cellular Respiration | Includes both aerobic and anaerobic processes |  | 55 |
| 1822557040 | Redox Reactions | A chemical reaction involving the complete or partial transfer of one or more electrons from one reactant to another; short for reduction-oxidation reaction |  | 56 |
| 1822557041 | Oxidation | The complete or partial loss of electrons from a substance involved in a redox reaction |  | 57 |
| 1822557042 | Reduction | The complete or partial addition of electrons to a substance involved in a redox reaction |  | 58 |
| 1822557043 | Reducing Agent | The electron donor in a redox reaction |  | 59 |
| 1822557044 | Oxidizing Agent | The electron receptor in a redox reaction |  | 60 |
| 1822557045 | NAD+ | Nicotinamide adenine dinucleotide, a coenzyme that cycles easily between oxidized (NAD+) and reduced (NADH) states, thus acting as an electron carrier. |  | 61 |
| 1822557046 | Electron Transport Chain | A sequence of electron carrier molecules (membrane proteins) that shuttle electrons down a series of redox reactions that release energy used to make ATP |  | 62 |
| 1822557047 | Glycolysis | A series of reactions that ultimately splits glucose into pyruvate. Occurs in almost all living cells, serving as the starting point for fermentation or cellular respiration |  | 63 |
| 1822556959 | The Citric Acid Cycle | A chemical cycle involving eight steps taht completes the metabolic breakdown of glucose molecules begun in glycolysis by oxidyzing acetyl CoA (derived from pyruvate) to carbon diozide; occcurs within the mitochondrion in eukaryotic cells and in the cytosol of prokaryotes; together with pyruvate oxidation, the second majoy stage in cellular respiration. | | 64 |
| 1822557048 | Oxidative Phosphorylation | The production of ATP using energy derived from the redox reactions of an electron transport chain; the third major stage of cellular respiration |  | 65 |
| 1822557049 | Substrate-level phosphorylation | The enzyme catalyzed formation of ATP by direct transfer of a phosphate group to ADP from an intermediate substrate in catabolism |  | 66 |
| 1822557050 | Acetyl CoA | Acetyl coenzyme A; the entry compound for the citric acid cycle in cellular respiration, formed from a fragment of pyruvate attached to a coenzyme. |  | 67 |
| 1822557051 | Cytochromes | An iron-containing protein that is a component of electron transport chains in the mitochondria and chloroplasts of eukaryotic cells and the plasma membranes of prokaryotic cells |  | 68 |
| 1822557052 | ATP Synthase | A complex of several membrane proteins that functions in chemiosmosis with adjacent electron transport chains, using the energy of a hydrogen ion (proton) concentration gradient to make ATP. ATP synthases are found in the inner mitochondrial membranes of eukaryotic cells and in the plasma membranes of prokaryotes. |  | 69 |
| 1822557053 | Chemiosmosis | An energy coupling mechanism that uses energy stored in the form of a hydrogen ion gradient across a membrane to drive cellular work; such as the synthesis of ATP. Under aerobic conditions, most ATP synthesis in cells occurs by chemiosmosis. |  | 70 |
| 1822557054 | Protonmotive Force | The potential energy stored in the form of a proton electrochemical gradient, generated by the pumping of hydrogen ions (H+) across a biological membrane during chemiosmosis. |  | 71 |
| 1822557055 | Alcohol Fermentation | Glycolysis followed by the reduction pyruvate to ethyl alcohol, regenerating NAD+ and releasing carbon dioxide |  | 72 |
| 1822557056 | Lactic Acid Fermentation | Glycolysis followed by the reduction of pyruvate to lactate, regenerating NAD+ with no release of of carbon dioxide. |  | 73 |
| 1822557057 | Obligate Anaerobes | An organism that only carries out fermentation or anaerobic respiration. cannot use oxygen and may be poisoned by it |  | 74 |
| 1822557058 | Facultative Anaerobes | An organism that makes ATP by aerobic respiration if oxygen is present but that switches to anaerobic respiration or fermentation if oxygen is not present |  | 75 |
| 1822557059 | Beta Oxidation | A metabolic sequence that breaks fatty acids down to two-carbon fragments that enter the citric acid cycle as acetyl CoA |  | 76 |
| 1822556960 | In the cytosol | in cellular respiration, where does Glycolysis occur? | | 77 |
| 1822557060 | What is pyruvate? | - The end product of glycolysis
-The first step in cellular respiration
- Stands in the junction between anaerobic and aerobic pathways |  | 78 |
| 1822556961 | The Citric Acid Cycle also known as the Krebs cycle serves what purpose? | ... | | 79 |
| 1822556962 | OIL RIG | Oxidation is Loss of Electrons (OIL)
RIG (Reduction is a Gain of Electrons) | | 80 |
| 1822556963 | Which process or processes return carbon to the atmosphere? | Cellular Respiration | | 81 |
| 1822556964 | What are the 4 major steps of cellular respiration? | 1.Glycolysis- (occurs in the cytosol)
2. Pyruvate Oxidation - Formation of Acetyl Co-enzyme A (occurs in the mitochondrion)
3. TCA/Kreb's Cycle/ Citric Acid Cycle
4. Oxidative phosphorylation - Electron Transport Chain and Chemiosmosis - (occurs in the inner membrane of the mitochondrion) | | 82 |
| 1822556965 | In the Cellular Respiration Process, which coenzymes is an electron carrier? | NADH and FADH2 | | 83 |
| 1822556966 | How many ATP are produced by the citric acid cycle for each molecule of glucose? | 2 | | 84 |
| 1822556967 | What is the correct sequence of steps as energy is extracted from glucose during cellular respiration. | glycolysis → acetyl CoA → citric acid cycle → electron transport chain | | 85 |
| 1822556968 | What is the correct general equation for cellular respiration? | C6H12O6 + 6 O2 → 6 CO2 + 6 H2O + ATP energy | | 86 |
| 1822556969 | What is the correct basic equation for cellular respiration? | Glucose + 6 Oxygen leads to 6 Carbon Dioxide +6 Water + 1 ATP | | 87 |
| 1822556970 | Which of the part of the cellular respiration process takes place in the cytosol of a eukaryotic cell? | Glycolysis | | 88 |
| 1822556971 | In what organelle would you find acetyl CoA formation, the citric acid cycle, and the electron transport chain? | The Mitochondrion | | 89 |
| 1822556972 | How would you describe glycolysis? | This process splits glucose in half and produces 2 ATPs for each glucose. | | 90 |
| 1822556973 | How would you describe the citric acid cycle | This process produces some ATP and carbon dioxide in the mitochondrion. | | 91 |
| 1822556974 | How would you describe the electron transport chain? | This process uses energy captured from electrons flowing to oxygen to produce most of the ATPs in cellular respiration | | 92 |
| 1822556975 | Which part of a eukaryotic cell is involved in cellular respiration? | Mitochondria | | 93 |
| 1822556976 | Glucose and _______ are consumed during cellular respiration | Oxygen | | 94 |
| 1822556977 | In a eukaryotic cell, most of the ATP derived from glucose during cellular respiration is produced by ... | The Electron Transport Chain | | 95 |
| 1822556978 | How much does a single glucose molecule produce in glycoloysis alone? | a single glucose molecule in glycolysis produces a total of:
2 molecules of pyruvic acid,
2 molecules of ATP,
2 molecules of NADH and
2 molecules of water | | 96 |
| 1822556979 | The immediate energy source that drives ATP synthesis by ATP synthase during oxidative phosphorylation is the | H+ concentration across the membrane holding ATP synthase. | | 97 |
| 1822556980 | Which metabolic pathway is common to both fermentation and cellular respiration of a glucose molecule? | glycolysis | | 98 |
| 1822556981 | In mitochondria, exergonic redox reactions | provide the energy that establishes the proton gradient. | | 99 |
| 1822556982 | The final electron acceptor of the electron transport chain that functions in aerobic oxidative phosphorylation is | oxygen. | | 100 |
| 1822556983 | What is the oxidizing agent in the following reaction?
Pyruvate + NADH + H+ S Lactate + NAD+ | pyruvate | | 101 |
| 1822556984 | When electrons flow along the electron transport chains of mitochondria, which of the following changes occurs?
ATP synthase pumps protons by active transport.
The pH of the matrix increases. | The pH of the matrix increases. | | 102 |
| 1822556985 | Most CO2 from catabolism is released during | the citric acid cycle. | | 103 |
| 1822556986 | A molecule becomes more oxidized when it _ | Loses an electron. During oxidation, an electron is removed. | | 104 |
| 1822556987 | In the overall process of glycolysis and cellular respiration, _____ is oxidized and _____ is reduced. | glucose ... oxygen
In cellular respiration, electrons and hydrogen are removed from glucose and added to oxygen. | | 105 |
| 1822556988 | Most of the ATP produced in cellular respiration comes from which of the following processes? | ... | | 106 |
| 1822556989 | Which of the following is a correct description of the events of cellular respiration and the sequence of events in cellular respiration? (eText Concept 9.1)
oxidation of glucose to pyruvate; reduction of pyruvate; TCA cycle; oxidative phosphorylation
glycolysis; oxidative phosphorylation; TCA cycle; oxidation of pyruvate.
oxidation of glucose to pyruvate; oxidation of pyruvate; oxidation of acetyl-coA; oxidative phosphorylation
oxidation of pyruvate; TCA cycle; oxidation of glucose to pyruvate; oxidative phosphorylation
glycolysis; reduction of pyruvate; TCA cycle; oxidative phosphorylation | 1. oxidation of glucose to pyruvate;
2. oxidation of pyruvate;
3. oxidation of acetyl-coA;
4. oxidative phosphorylation | | 107 |
| 1822556990 | Oxygen gas (O2) is one of the strongest oxidizing agents known. The explanation for this is that _____. (eText Concept 9.1)
oxygen gas contains a double bond
oxygen acts as the final electron acceptor in cellular respiration
the oxygen atom is very electronegative
oxygen is so abundant in the atmosphere
oxygen gas is composed of two atoms of oxygen | The oxygen atom is very electronegative.
The high electronegativity of the oxygen atom is the reason that oxygen is such a good oxidizing agent (why it can oxidize so many compounds). | | 108 |
| 1822556991 | The function of cellular respiration is to _____. (eText Concept 9.1)
reduce CO2
produce carbohydrates
extract CO2 from the atmosphere
synthesize macromolecules from monomers
extract usable energy from glucose | Extract usable energy from glucose.
The most prevalent and efficient catabolic pathway is cellular respiration, in which oxygen is consumed as a reactant, along with the organic fuel (frequently glucose). | | 109 |
| 1822556992 | During the reaction C6H12O6 + 6 O2 → 6 CO2 + 6 H2O, which compound is reduced as a result of the reaction? (eText Concept 9.1)
carbon dioxide
oxygen
water
glucose
both glucose and carbon dioxide | oxygen | | 110 |
| 1822556993 | Each ATP molecule contains about 1% of the amount of chemical energy available from the complete oxidation of a single glucose molecule. Cellular respiration produces about 32 ATP from one glucose molecule. What happens to the rest of the energy in glucose? (eText Concept 9.1)
It is converted to starch.
It is converted to heat.
It is released as carbon dioxide and water
It is stored as fat.
It is used to make water from hydrogen ions and oxygen. | It is converted to heat. | | 111 |
| 1822556994 | A small amount of ATP is made in glycolysis by which of the following processes? (eText Concept 9.1)
transfer of electrons and hydrogen atoms to NAD+
transport of electrons through a series of carriers
attachment of a free inorganic phosphate (Pi) group to ADP to make ATP
transfer of a phosphate group from a fragment of glucose to ADP by substrate-level phosphorylation
harnessing energy from the sun | Transfer of a phosphate group from a fragment of glucose to ADP by substrate-level phosphorylation | | 112 |
| 1822556995 | Where do the reactions of glycolysis occur in a eukaryotic cell? (eText Concept 9.1)
the cytosol
across the inner membrane of the mitochondrion
the matrix of the mitochondrion
in the intermembrane space of the mitochondrion
the inner membrane of the mitochondrion | Glycolysis occurs in the cytosol. | | 113 |
| 1822556996 | Most of the NADH that delivers electrons to the electron transport chain comes from which of the following processes? (eText Concept 9.3)
substrate-level phosphorylation
the citric acid cycle
glycolysis
anabolic pathways
oxidative phosphorylation | The citric acid cycle
Both NADH and FADH2 are produced during the citric acid cycle. | | 114 |
| 1822556997 | In preparing pyruvate to enter the citric acid cycle, which of the following steps occurs? (eText Concept 9.3)
Pyruvate is oxidized and decarboxylated, and the removed electrons are used to reduce an NAD+ to an NADH.
Pyruvate is reduced and decarboxylated, and the resulting electrons oxidize an NAD+ to an NADH
Pyruvate is oxidized and decarboxylated, and the resulting electrons are donated to NADH to produce NAD+.
Pyruvate is reduced to acetyl-coA, which involves the reduction of pyruvate, the addition of a carbon dioxide from the environment, and its reduction by NADH.
Pyruvate is ionized directly to acetyl-coA. | Pyruvate is oxidized and decarboxylated, and the removed electrons are used to reduce an NAD+ to an NADH. | | 115 |
| 1822556998 | Why is the citric acid cycle called a cycle? (eText Concept 9.3) | The four-carbon acid that accepts the acetyl CoA in the first step of the cycle is regenerated by the last step of the cycle. | | 116 |
| 1822556999 | In the citric acid cycle, for each pyruvate that enters the cycle, one ATP, three NADH, and one FADH2 are produced. For each glucose molecule that enters glycolysis, how many ATP, NADH, and FADH2 are produced in the citric acid cycle? (eText Concept 9.3) | two ATP, six NADH, two FADH2 | | 117 |
| 1822557000 | Where do the reactions of the citric acid cycle occur in eukaryotic cells? | The matrix of the mitochondrion
The citric acid cycle, which takes place in the mitochondrial matrix, completes the degradation of glucose. | | 118 |
| 1822557001 | How many molecules of ATP are gained by substrate-level phosphorylation from the complete breakdown of a single molecule of glucose in the presence of oxygen? (eText Concept 9.3) | Four
There is a net gain of two ATP from glycolysis and one per each molecule of acetyl CoA oxidized in the citric acid cycle for a total of four per glucose. | | 119 |
| 1822557002 | What is the major (but not the only) energy accomplishment of the citric acid cycle? (eText Concept 9.3) | The formation of NADH and FADH2
Each turn of the citric acid cycle releases carbon dioxide, forms one ATP by oxidative phosphorylation, and passes electrons to three NAD+ and one FAD. | | 120 |
| 1822557003 | After completion of the citric acid cycle, most of the usable energy from the original glucose molecule is in the form of _____. | NADH
The citric acid cycle produces three NADH per turn of the cycle. | | 121 |
| 1822557004 | Which of the following accompanies the conversion of pyruvate to acetyl CoA before the citric acid cycle? (eText Concept 9.3)
regeneration of NAD+
removal of coenzyme A
release of CO2 and synthesis of NADH
formation of CO2 and synthesis of ATP
release of CO2 and release of coenzyme A | release of CO2 and synthesis of NADH | | 122 |
| 1822557005 | If significant amounts of materials are removed from the citric acid cycle to produce amino acids for protein synthesis, which of the following will result? (eText Concept 9.6)
Less ATP will be produced by the cell.
Less CO2 will be produced by the cell.
The four-carbon compound that combines with acetyl CoA will have to be made by some other process.
The first two answers are correct.
The first three answers are correct. | The first three answers are correct. | | 123 |
| 1822557006 | A gram of fat oxidized by respiration produces approximately twice as much ATP as a gram of carbohydrate. Which of the following best explains this observation? (eText Concept 9.6)
Fats are better electron donors to oxygen than are sugars.
Fats do not form true macromolecules as sugars do.
Fats are closely related to lipid molecules, the basic building blocks of cellular membranes.
Fats are less soluble in water than sugars.
Fats are produced when cells take in more food than they need. | Fats are better electron donors to oxygen than are sugars.
Correct. Fats contain more hydrogen and less oxygen than sugars—it is the transfer of electrons from good donors such as hydrogen atoms to good acceptors such as oxygen that provides the energy in respiration. | | 124 |
| 1822557007 | When protein molecules are used as fuel for cellular respiration, _____ are produced as waste. (eText Concept 9.6)
ethanol and CO2
molecules of lactate
fatty acids
amino groups
sugar molecules | amino groups
The amino group is a residual product of amino acid catabolism. | | 125 |
| 1822557008 | Of the metabolic pathways listed below, which is the only pathway found in all organisms? (eText Concept 9.5)
the electron transport chain
cellular respiration
fermentation
the citric acid cycle
glycolysis | glycolysis
Ancient prokaryotes probably used glycolysis to make ATP long before oxygen was present in Earth's atmosphere. Glycolysis is the most widespread metabolic pathway. | | 126 |
| 1822557009 | If muscle cells in the human body consume O2 faster than it can be supplied, which of the following is likely to result? (eText Concept 9.5)
The muscle cells will have more trouble making enough ATP to meet their energy requirements.
The cells will not be able to carry out oxidative phosphorylation.
The cells will consume glucose at an increased rate.
Only the first two answers are correct.
The first three answers are correct. | The first three answers are correct.
The muscle cells will have more trouble making enough ATP to meet their energy requirements.
The cells will not be able to carry out oxidative phosphorylation.
The cells will consume glucose at an increased rate. | | 127 |
| 1822557010 | In brewing beer, maltose (a disaccharide of glucose) is _____. | the substrate for alcoholic fermentation | | 128 |
| 1822557011 | In glycolysis in the absence of oxygen, cells need a way to regenerate which compound? (eText Concept 9.5) | NAD+ | | 129 |
| 1822557012 | Sports physiologists at an Olympic training center wanted to monitor athletes to determine at what point their muscles were functioning anaerobically. They could do this by checking for a buildup of which of the following compounds? (eText Concept 9.5) | lactate.
In humans, muscle cells switch to lactate fermentation after becoming anaerobic. | | 130 |
| 1822557013 | Fermentation is essentially glycolysis plus an extra step in which pyruvate is reduced to form lactate or alcohol and carbon dioxide. This last step _____. (eText Concept 9.5) | enables the cell to recycle the reduced NADH to oxidized NAD+ | | 131 |
| 1822557014 | During respiration in eukaryotic cells, the electron transport chain is located in or on the _____. (eText Concept 9.4) | inner membrane of the mitochondrion | | 132 |
| 1822557015 | Which of the following is the source of the energy that produces the chemiosmotic gradient in mitochondria? (eText Concept 9.4) | Electrons moving down the electron transport chain | | 133 |
| 1822557016 | Which of the following best describes the electron transport chain? (eText Concept 9.4)
Acetyl CoA is fully oxidized to CO2.
Glucose is broken down to a three-carbon compound in preparation for the citric acid cycle.
Electrons are passed from one carrier to another, releasing a little energy at each step.
Hydrogen atoms are added to CO2 to make an energy-rich compound.
Electrons are pumped across a membrane by active transport. | Electrons are passed from one carrier to another, releasing a little energy at each step. | | 134 |
| 1822557017 | During aerobic respiration, molecular oxygen (O2) is used for which of the following purposes? (eText Concept 9.4)
at the end of glycolysis to oxidize pyruvate
at the end of the electron transport chain to accept electrons and form H2O
between glycolysis and the citric acid cycle to split a carbon from pyruvate, producing CO2
as a source of O2 in every reaction that produces CO2
at the end of the citric acid cycle to regenerate citric acid | at the end of the electron transport chain to accept electrons and form H2O | | 135 |
| 1822557018 | Word Roots: aero- | air (aerobic: chemical reaction using oxygen) | | 136 |
| 1822557019 | Word Roots: an- | not (anaerobic: chemical reaction not using oxygen) | | 137 |
| 1822557020 | Word Roots: chemi- | chemical (chemiosmosis: the production of ATP using the energy of hydrogen ion gradients across membranes to phosphorylate ADP) | | 138 |
| 1822557021 | Word Roots: glyco- | sweet; | | 139 |
| 1822557022 | Word Roots: -lysis | split (glycolysis: the splitting of glucose into pyruvate) | | 140 |

















































































































